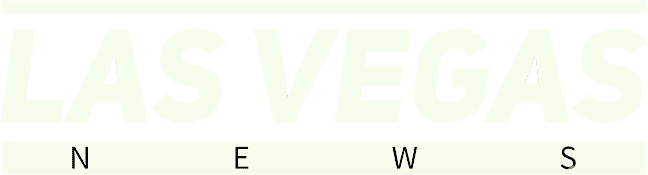Science has always been humanity’s most reliable compass, pointing toward longer lives, deeper understanding, and a more just world. Some discoveries reshape a single field; others rewrite the rules of everything we thought we knew. The breakthroughs highlighted here span gene editing, brain mapping, protein prediction, HIV prevention, and the nanoscale architecture of the human brain. Each one, in its own way, has nudged civilization forward – permanently.
1. CRISPR Gene Editing: Rewriting the Code of Life

CRISPR, short for Clustered Regularly Interspaced Short Palindromic Repeats, is the foundation of modern gene-editing technology. It allows scientists to locate specific DNA sequences and make targeted changes, often replacing faulty genetic code with healthy versions. The system is based on a natural defense mechanism found in bacteria, which use CRISPR to recognize and cut up the DNA of invading viruses. What began as a quirk of bacterial immunity has since evolved into one of the most powerful molecular tools ever placed in human hands. The pace of clinical progress is now extraordinary, with trials running on multiple continents for dozens of conditions.
The first approved CRISPR drug, Casgevy, edits genes in patients’ stem cells by using a mixture of guide RNA and Cas9 RNP in vitro, inducing the expression of fetal hemoglobin to compensate for the lack of adult hemoglobin function in patients with sickle cell disease. During the past year, Casgevy has been approved in the UK, US, EU, Bahrain, Saudi Arabia, and Canada. Beyond blood disorders, the technology is moving into cardiology. Results from a Cleveland Clinic Phase 1, first-in-human clinical trial showed that a one-time infusion of a gene-editing therapy using CRISPR-Cas9 safely reduced LDL cholesterol and triglycerides in people with lipid disorders resistant to current medications. The results were presented at the American Heart Association’s Scientific Sessions 2025 and simultaneously published in the New England Journal of Medicine. In the trial, which included 15 patients, both LDL cholesterol and triglyceride levels were substantially reduced within two weeks after treatment and stayed at low levels for at least 60 days.
2. Lenacapavir: A Near-Perfect Shield Against HIV

Lenacapavir, a long-acting injectable pre-exposure prophylaxis taken once every six months, demonstrated 100% protection against HIV infections among cisgender women enrolled in the PURPOSE 1 clinical trial conducted in South Africa and Uganda, researchers presented at AIDS 2024, the 25th International AIDS Conference. The results were so extraordinary that the trial’s independent Data Monitoring Committee recommended stopping the blinded phase immediately and offering the drug to all participants. Since then, hundreds of trial participants opted to initiate lenacapavir, highlighting the desire for a long-acting injectable PrEP option compared to daily oral PrEP.
The PURPOSE 1 trial found 100% efficacy in preventing HIV in 5,300 cisgender women in Uganda and South Africa, and the PURPOSE 2 trial showed a 96% reduction in HIV incidence among cisgender men, trans, and non-binary individuals across multiple countries. Both trials demonstrated lenacapavir’s safety and effectiveness in reducing HIV transmission. In June 2025, the FDA approved injectable lenacapavir for PrEP. The journal Science named lenacapavir its 2024 “Breakthrough of the Year.” The drug’s mechanism is equally remarkable: the success of lenacapavir as PrEP sprang from a basic research advance – a new understanding of the structure and function of HIV’s capsid protein, which it targets. Many other viruses have their own capsid proteins, which form a shell around their genetic material, so this drug’s triumph raises the prospect that similar capsid inhibitors could fight other viral diseases.
3. AlphaFold: Solving Biology’s Greatest Puzzle
Hassabis and Jumper created AlphaFold, which in 2020 solved a problem scientists had been wrestling with for decades: predicting the three-dimensional structure of a protein from a sequence of amino acids. For context, figuring out a single protein’s shape using traditional laboratory methods could take years and hundreds of thousands of dollars. AlphaFold changed that overnight. In 2020, Hassabis and Jumper developed AlphaFold2, which can predict the three-dimensional structure of a protein, using only the sequence of letters, with over 90% accuracy.
Demis Hassabis and John Jumper shared one half of the 2024 Nobel Prize in Chemistry, awarded “for protein structure prediction,” while the other half went to David Baker “for computational protein design.” The scale of adoption is staggering. AlphaFold is being used by over 3 million researchers from over 190 countries around the world, tackling problems such as antimicrobial resistance, crop resilience, and heart disease. AlphaFold 3 was announced on 8 May 2024. It can predict the structure of complexes created by proteins with DNA, RNA, various ligands, and ions. The new prediction method shows a minimum 50% improvement in accuracy for protein interactions with other molecules compared to existing methods.
4. Mapping the Brain’s Wiring: The Complete Fruit Fly Connectome

A scientific team supported by the National Institutes of Health unveiled the first complete map of the neural connections of the common fruit fly brain. The map provides a wiring diagram, known as a connectome, and is the largest and most complete connectome of an adult animal ever created. This work offers critical information about how brains are wired and the signals that underlie healthy brain functions. The study details over 50 million connections between more than 130,000 neurons, appearing as part of a package of nine papers in the journal Nature. The effort began in 2018 and involved hundreds of researchers, artificial intelligence tools, and volunteer citizen scientists from around the world.
Adult fruit flies share 60 percent of human DNA, as well as 75 percent of the genes that cause genetic diseases. Understanding the fly’s brain in such detail could hold implications for connections in human brains and the neural pathways that lead to certain behaviors. For many heartbreaking diseases of the brain – dementia, Parkinson’s, Alzheimer’s and others – doctors can only treat the symptoms. Medical science does not have a cure. The connectome is a step toward changing that reality. Tools developed for FlyWire could also be used to analyze more complex brains in the same way, setting the stage for eventual mapping of the mouse, zebrafish, and ultimately human brains.
5. Mapping the Human Brain at Nanoscale Resolution
A cubic millimetre of the human brain was mapped at nanoscale resolution by a team at Google. This contains roughly 57,000 cells and 150 million synapses, incorporating 1.4 petabytes of data. To put that into perspective, a cubic millimeter is roughly the size of a grain of sand – yet the data it contained was so vast it required a petabyte of storage to hold. This achievement, published in 2024, marked a historic milestone: for the first time, scientists had charted a slice of the human brain in precise, atom-scale detail, revealing cellular architecture never seen before.
The connectome charts the wiring between neurons, giving new insights into the roles of different brain regions. The human brain presents a far bigger challenge, with more than 80 billion neurons. Still, the nanoscale map of even a tiny portion of human brain tissue is opening entirely new questions about how memory is stored and how consciousness arises. Artificial intelligence algorithms are helping to tackle these data-heavy tasks. Along with seeking to better understand fundamentals of how the brain works, such projects aim to solve mysteries of human mental illnesses or chart better paths to recovery after brain injury – and, along the way, might also help to improve AI. The line between neuroscience and artificial intelligence is becoming thinner with every new discovery.
6. CRISPR Without Cutting: The Next Frontier of Gene Therapy
A new CRISPR breakthrough shows scientists can turn genes back on without cutting DNA, by removing chemical tags that act like molecular anchors. The work confirms these tags actively silence genes, settling a long-running scientific debate. This gentler form of gene editing could offer a safer way to treat sickle cell disease by reactivating a fetal blood gene. Researchers say it opens the door to powerful therapies with fewer unintended side effects. This approach, known as epigenetic editing, sidesteps one of the original technology’s biggest risks – that cutting DNA could trigger unintended mutations elsewhere in the genome.
Scientists at UNSW Sydney developed this new form of CRISPR technology that could make gene therapy safer while also resolving a decades-long debate about how genes are switched off. The research shows that small chemical markers attached to DNA actively silence genes, rather than simply appearing as harmless byproducts in inactive regions of the genome. New trends to watch include the accelerated expansion of green chemistry, the clinical validation of CRISPR, the rise of biomaterials, and renewed progress in treating the undruggable, from cancer to neurodegenerative diseases. The era of epigenetic medicine is no longer theoretical. Published in Nature Communications in January 2026, this UNSW finding represents one of the most significant refinements to CRISPR technology since its original inception, and could redefine how genetic disease is treated for generations to come.