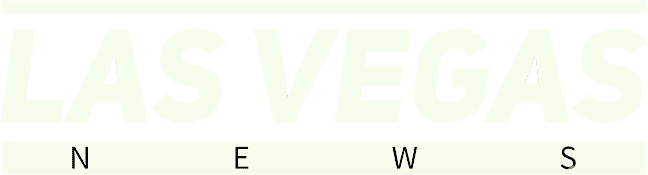We are living through one of the most dramatic moments in medical history. The obesity drug market is not just evolving. It is exploding. What started with weekly injections in your fridge has turned into a full-scale pharmaceutical revolution, with pills, monthly shots, triple-hormone combinations, and drugs that may actually rival bariatric surgery in terms of results.
The race is on. Dozens of new compounds are entering the field, each promising more weight loss, fewer side effects, or easier dosing. Some of these are already approved and hitting pharmacy shelves right now. Others are so close to the finish line, you can almost touch them. Let’s dive in.
1. The Wegovy Pill: The Shot That Became a Swallow
In December 2025, the FDA approved the Wegovy pill to reduce excess body weight and maintain weight reduction long term, making it the first oral GLP-1 to reach the market. That is a genuinely huge deal. For millions of people who dreaded weekly injections or had access issues, this changes everything.
Novo Nordisk announced that the FDA approved the Wegovy pill – once-daily oral semaglutide 25 mg – making it the first oral GLP-1 receptor agonist therapy approved for weight management. Honestly, getting semaglutide into pill form has been a scientific challenge for years. The stomach is not kind to peptides, which makes this a genuine chemistry feat.
In the OASIS 4 trial, oral semaglutide 25 mg taken once daily demonstrated 16.6% mean weight loss when treatment was adhered to in adult participants with obesity or overweight with one or more comorbidities. Starting in early January, the starting dose of 1.5 milligrams became available in pharmacies and via select telehealth providers with savings offers for $149 per month.
2. Foundayo (Orforglipron): Eli Lilly’s Pill That Plays by Different Rules

The orforglipron pill, called Foundayo, is made by Eli Lilly, which also sells the GLP-1 drugs Zepbound for obesity and Mounjaro for diabetes – both given as weekly injections – which along with rival drugs Wegovy and Ozempic have transformed weight-loss and diabetes treatment. Now Lilly is taking their shot at the pill market too. This just received FDA approval at the start of April 2026.
Lilly says Foundayo represents an advance because, unlike the Wegovy pill, it can be taken at any time of day and without restrictions on food and water. That sounds minor until you realize how many people struggle with the strict fasting requirements of other oral GLP-1s. Think of it like the difference between a supplement that demands an empty stomach versus one you can take with your morning coffee.
Orforglipron is a small-molecule nonpeptide oral GLP-1 inhibitor that completed a phase 3 clinical trial in 2025. Small-molecule nonpeptide GLP-1 RAs are more stable in the stomach and can be taken with food, and phase 3 results showed an 11.2% weight loss at the 36 mg dose at 72 weeks. It’s not the biggest number in the pipeline, but paired with its convenience, the commercial case is strong.
3. Retatrutide: The Triple Threat That Could Rewrite Everything

Retatrutide is an investigational once-weekly triple hormone receptor agonist – a single molecule that activates the body’s receptors for GIP, GLP-1, and glucagon. Three hormones. One shot. That is an entirely new category of drug, not merely a tweak to what already exists.
Lilly said in December that in a Phase 3 clinical trial, patients taking the highest dose of the experimental injection lost nearly 29% of their body weight on average after about 16 months – more than has been seen with any GLP-1 drug currently on the market. To put that in perspective: for someone weighing 250 pounds, that is roughly 72 pounds lost. Numbers like that were previously reserved for gastric bypass surgery.
Some trial participants slimmed down so dramatically that they stopped taking retatrutide over “perceived excessive weight loss,” Eli Lilly noted in the news release. As of April 2, 2026, no New Drug Application for retatrutide has been publicly confirmed, meaning FDA approval is most likely still a year or two away. Still, the science here is breathtaking.
4. CagriSema: Novo Nordisk’s Powerful Double Combination

A combination of an amylin analog, cagrilintide, and semaglutide, known as CagriSema, is a single weekly injection under development by Novo Nordisk, with phase 3a results published in 2025. This is not just a next-generation drug – it is two proven concepts fused into one injection, designed to work on hunger from multiple angles at once.
CagriSema demonstrated meaningful weight-loss effects in two phase 3 studies. After about 68 weeks, adults without diabetes lost about 20% of their body weight on average, while adults living with type 2 diabetes saw about 14% weight loss along with improvements in blood sugar readings.
CagriSema’s manufacturer, Novo Nordisk, applied for the medication’s FDA approval in December 2025, and an approval decision will likely come sometime in 2026. A recent meta-analysis presented at an American cardiology conference found that CagriSema led to greater weight loss than its individual ingredients alone, and also led to greater improvements in blood sugar, waist circumference, and blood pressure.
5. MariTide: Amgen’s Monthly Shot With a Long Half-Life

In a Phase II trial, Amgen’s investigational therapy MariTide delivered significant, sustained weight loss of up to 20% in adults with obesity, with or without type 2 diabetes, alongside notable improvements in glycemic control, supporting its advancement to Phase III studies as a potential once-monthly treatment option. Monthly. Not weekly. That is a potentially game-changing dosing advantage for people who struggle with adherence.
Maridebart cafraglutide is a long-acting peptide-antibody conjugate that combines GLP-1 receptor agonism and GIP receptor antagonism, with a 21-day half-life – three times longer than the FDA-approved longest-acting once-weekly anti-obesity medication, semaglutide. That extended biological lifespan is what enables the monthly dosing schedule, which is genuinely novel in this space.
A weight plateau was not reached at 52 weeks, with weight continuing a downward trajectory, meaning longer-term trials are needed to assess the full weight efficacy of this agent. The recently initiated Phase 3, 72-week chronic weight management studies will evaluate the safety, efficacy and tolerability of MariTide in participants living with obesity or overweight with and without Type 2 diabetes. The jury is still out, but the trajectory is promising.
6. Amycretin: A Daily Pill Targeting Two Hunger Hormones at Once

Amycretin is an oral medication being developed for chronic weight management. It’s a dual GLP-1 and amylin receptor agonist – and amylin is a hormone that helps you feel more full after meals. So rather than targeting just one satiety signal, amycretin essentially hits two of the brain’s hunger-off switches simultaneously.
Amycretin is a once-daily oral tablet, though an under-the-skin amycretin injection is also being developed. The ability to take it as a pill is significant, particularly for patients who want options beyond needles. Novo Nordisk is behind this one, and they have significant manufacturing and regulatory experience to push it through.
Amycretin is advancing into phase 3 studies, and an executive at Novo Nordisk predicted that amycretin will likely be available sometime this decade, following early studies involving people who are considered obese that showed significant and lasting weight loss. It’s hard to say for sure when it will land, but the momentum is real.
7. A Quadruple-Action Compound: Surgery-Level Weight Loss in a Drug
Tufts University chemists have created a new multi-target compound that goes beyond the usual GLP-1, GIP, and glucagon approaches by adding a fourth hormone, PYY. That’s right – four hormones. If the triple-agonist approach that powers retatrutide seems ambitious, this pushes the science even further.
Tufts University chemists created this compound with a “quadruple-action” design that aims to deliver weight loss results on par with bariatric surgery – up to 30% – without invasive procedures, and could change the future of obesity treatment. Let’s be real: achieving surgery-level outcomes without actually going under the knife would be a seismic shift in how medicine handles severe obesity.
This compound is still in very early development, so patients should temper their expectations. It will not be on pharmacy shelves anytime soon. Still, the science behind it signals where the field is heading, and that direction is remarkable. Think of it as a preview of what metabolic medicine might look like a decade from now.
8. GLP-1s Go Beyond Weight: A New Label for These Drugs

Dr. Peter Balazs, a hormone and weight loss specialist in New York and New Jersey, shared that the most important shift is likely to label GLP-1 drugs as “multi-system metabolic modulators” rather than “simple weight loss drugs,” noting that the treatment goal is no longer just BMI reduction, but total cardiometabolic risk mitigation, with effects now documented across the liver, heart, kidneys and vasculature.
Research has also shown a growing relationship between GLP-1 drugs and addiction. It started because, anecdotally, physicians were starting to see that their patients were reporting they hadn’t smoked in a while, or hadn’t had alcohol in a while. This was not an expected benefit – it emerged from real clinical observations and has since sparked a wave of formal research.
Several GLP-1s for obesity have been approved for new indications such as secondary cardiovascular risk reduction, sleep apnea and metabolic dysfunction-associated steatohepatitis. The idea that one drug class can touch cardiovascular disease, fatty liver, sleep apnea, and potentially addiction is not just exciting – it is genuinely unprecedented in the history of metabolic medicine.
9. The Market Behind the Medicine: A Trillion-Dollar Shift
Towards the end of 2025, estimates projected the global anti-obesity drugs market to grow from $19.6 billion in 2025 to $104.9 billion by 2035, representing a compound annual growth rate of 18.3%. That kind of growth curve usually only appears in technology sectors. The fact that it is happening in pharmaceuticals tells you everything about how serious the obesity epidemic has become.
Eli Lilly became the first pharmaceutical company to surpass a $1 trillion market capitalization, buoyed by global demand for obesity medications. Lilly’s first nine months of 2025 showed Mounjaro and Zepbound generating $39.5 billion in revenue, surpassing Merck’s Keytruda as the world’s best-selling medicine. That is a staggering commercial reality built in just a few years.
According to TD Cowen, there were over 160 obesity drugs in development in 2025, covering 68 mechanisms of action, including incretins, drugs that increase mitochondrial action, drugs that preserve muscle mass, and many other mechanisms. The pipeline is not just wide – it is deep. This field now attracts the kind of capital and talent that once went almost exclusively to oncology.
10. Access, Pricing, and What Comes Next

A study from FAIR Health published in May 2025 reports that more than 2% of U.S. adults took a GLP-1 for weight loss in 2024. That number sounds small until you realize how rapidly it is growing. Weight loss drugs like Ozempic and Wegovy are used by over 15 million adults in the U.S., or roughly 4.5% of the population. The mainstream adoption curve is steep.
On November 6, 2025, Novo Nordisk and Eli Lilly entered into a series of agreements with the U.S. government to expand access and lower the cost of GLP-1 agents for Medicare recipients beginning in 2026. Currently, medications for obesity alone are not covered under Medicare – and the new agreement will allow Medicare Part D coverage for anti-obesity medicines for certain populations. That is a policy shift that could bring these drugs to millions who have been priced out.
Amid the growth of artificial intelligence, experts are predicting an expansion in AI-driven weight-loss methods, including categorizing obesity into sub-types like “hungry brain,” “emotional hunger” and “slow burn” to personalize how therapy is prescribed while moving away from “trial and error.” The future is not just new molecules – it is smarter ways to match the right molecule to the right patient from the very start.
Conclusion: The Weight Loss Revolution Is Just Getting Started

Here’s the thing – Ozempic was not the end of this story. It was the opening chapter. The drugs now entering the pipeline are stronger, more flexible in how you take them, and increasingly targeted at the full range of consequences that come with metabolic disease. From pills you can take with breakfast, to monthly injections, to triple-hormone compounds that produce results previously only possible on an operating table, the landscape has fundamentally changed.
What is most remarkable is the speed. Five years ago, a drug producing roughly 15% weight loss was considered extraordinary. Today, nearly 30% weight loss in a clinical trial barely makes the front page. The science is running fast, and regulators, payers, and healthcare systems are scrambling to keep up.
The question worth sitting with is this: with so many options arriving so quickly, how do we make sure the right people get the right treatments – not just those who can afford them? What do you think? Tell us in the comments.